In case you are dealing with chemicals, drugs or materials—albeit indirectly—you most probably have accessed the PubChem database already, if not consciously. It is among the very few scientific resources that silently support everything from pharmaceutical R&D and toxicology dashboards to environmental health apps and even consumer-safety websites. PubChem is located at the crossroads of chemistry, biology, pharmacology, and open data, and it accomplishes an incredibly rare feat: it transforms the highly technical chemical information into a format that is understandable, searchable, and usable by anyone.
The article explains what PubChem is and how the professionals depend on it and how you can make use of it to take smarter, safer, and more informed decisions in research, industry, or even everyday life.
What Is the PubChem Database?
PubChem is the largest free chemistry database in the world, which is managed by the National Center for Biotechnology Information (NCBI) at the U.S. National Institutes of Health (NIH).
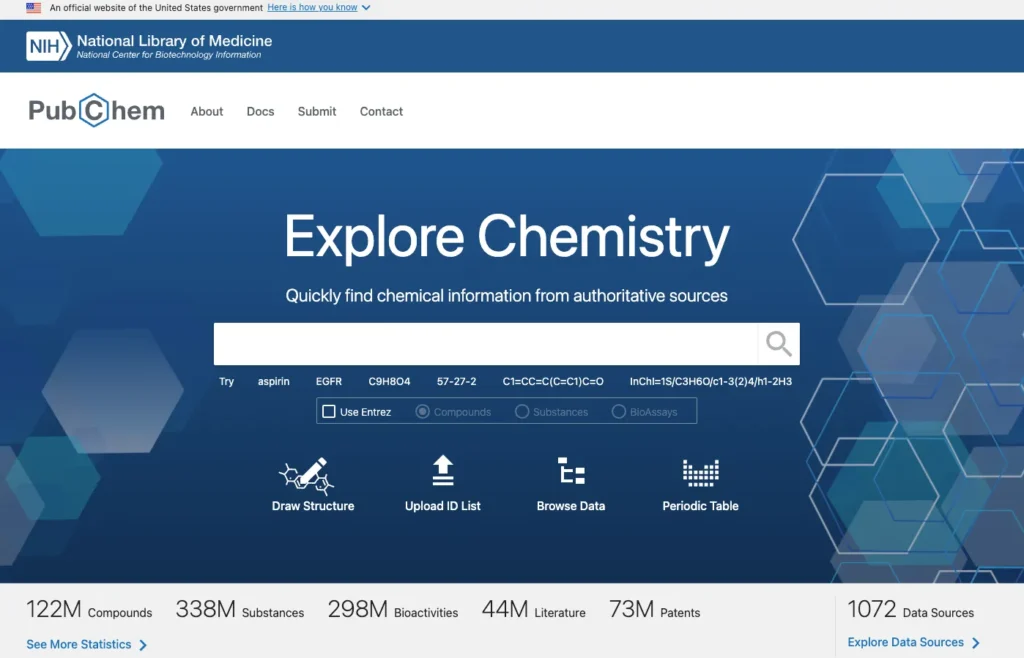
As per the official documentation of the project, more than 100 million chemical records are stored in PubChem containing various data types such as chemical structures, identifiers, safety and biological activity data, toxicology profiles, and references to thousands of scientific sources (PubChem About, NCBI).
The database is built for a very basic yet very powerful goal:
To make chemical information very open and accessible—very accurately, very transparently, and at very large scale.
The credibility of the database is based on the large number of reliable contributors: government agencies (like EPA, FDA, CDC), universities, pharmaceutical companies, environmental laboratories, and international research bodies.
Why the PubChem Database Matters (Even If You’re Not a Chemist)
PubChem’s value becomes obvious when you realize how many scientific questions hinge on knowing a single chemical’s structure or toxicity.
Consider these everyday use cases:
- Drug formulation scientists check drug–drug interactions and molecular pathways.
- Environmental researchers evaluate pesticides in water or air samples.
- Physicians and pharmacists verify medication risks and contraindications.
- Regulatory professionals review hazard classifications and exposure limits.
- Journalists investigate chemicals found in consumer products.
- Consumers look up toxicity information (e.g., “Is BPA dangerous?”).
Whenever you need to identify a substance, understand how it behaves, or determine whether it’s hazardous, PubChem is the authoritative starting point.
How the PubChem Database Is Structured: Three Core Record Types
PubChem organizes its information into three major record types:
1. Substance Records (SID)
Submitted by data providers such as labs, manufacturers, or agencies.
Useful for comparing how different organizations describe the same chemical.
2. Compound Records (CID)
Standardized structures created by PubChem by merging identical substances.
These are the most commonly viewed records.
3. BioAssay Records (AID)
Contain detailed test results on how chemicals behave in biological systems.
Together, these form a multi-layered dataset that supports both high-level browsing and advanced scientific analysis.
What You Can Find in a PubChem Database Record
(Using the Example of Acetaminophen – CID 1983)
To illustrate how deep PubChem’s data goes, let’s walk through a concrete example using a well-known compound: acetaminophen, the active ingredient in Tylenol.
When you search “acetaminophen” in PubChem, you’ll see tabs covering:
1. Structures
- 2D structural diagrams
- 3D conformers
- SMILES, InChI, and InChIKey identifiers
- Isomer and stereochemistry information
This is foundational for computational chemistry, drug design, and regulatory identification.
2. Names and Identifiers
Includes:
- Synonyms (e.g., paracetamol)
- CAS number
- UNII code
- EC and ECHA identifiers
This is especially valuable when comparing regulations across countries.
3. Chemical and Physical Properties
PubChem provides experimentally measured or predicted properties, such as:
- Molecular weight
- Melting point
- Boiling point
- LogP
- Solubility
- Vapor pressure
For acetaminophen, you’ll find its moderately high melting point and low vapor pressure—properties that influence formulation, stability, and environmental behavior.
4. Spectral Information
This is one of PubChem’s under-appreciated strengths.
You can access:
- NMR spectra
- IR spectra
- UV–Vis datasets
- Mass spectrometry profiles
Environmental chemists, forensic labs, and academic researchers rely on these to identify unknown samples.
5. Chemical Vendors
If you need to purchase a substance, PubChem lists verified suppliers, helping avoid counterfeit or mislabeled chemical products.
6. Drug and Medication Information
For pharmaceuticals, PubChem integrates:
- Mechanism of action
- Therapeutic uses
- Dosage forms
- Regulatory status
For acetaminophen, you’ll find FDA drug label references and links to the DailyMed database.
7. Pharmacology and Biochemistry
Includes:
- Metabolic pathways
- Target receptors or enzymes
- ADME profiles
- Bioactivity summaries
This data is critical for drug discovery and toxicology.
8. Safety and Hazards
PubChem imports or aggregates:
- OSHA classifications
- GHS hazard statements
- NFPA ratings
- Flammability, reactivity, instability data
- First-aid measures
- Fire-fighting instructions
This section alone makes PubChem indispensable for workplace safety programs.
9. Toxicity
Here you’ll find:
- Acute toxicity (LD50, LC50)
- Chronic toxicity
- Carcinogenicity, mutagenicity
- Reproductive toxicity
- Environmental toxicity (aquatic, terrestrial)
For acetaminophen, PubChem notes hepatotoxicity risks at high doses—reinforced by NIH and FDA references.
10. Associated Disorders and Diseases
PubChem links chemicals to:
- Related diseases
- Medical conditions
- Known adverse effects
For acetaminophen, this includes liver failure and overdose-related complications.
11. Literature and Patents
PubChem integrates:
- Peer-reviewed papers
- Clinical studies
- Patent documents from USPTO, EPO, WIPO
This allows researchers to trace a chemical’s development over decades.
12. Interactions and Pathways
PubChem connects chemicals to:
- Biological pathways (via KEGG, Reactome, etc.)
- Protein interactions
- Metabolic systems
For example, acetaminophen metabolism through CYP450 enzymes appears here with clear pathway visualizations.
13. Biological Test Results
These include results from NIH’s high-throughput screening programs and hundreds of bioassays.
Who Uses the PubChem Database—and Why It’s Become a Default Industry Tool
Pharmaceutical Companies
To validate compound identities, check pre-existing literature, and review toxicity.
Regulatory Agencies
EPA, FDA, ECHA, and OSHA rely on PubChem as a reference point for standard identifiers.
Environmental Scientists
To track contaminants, pesticides, industrial chemicals, and emerging pollutants.
Academics
For research, coursework, and experimental planning.
Consumer Safety Groups
To investigate chemicals used in cosmetics, food packaging, and household products.
Data Scientists
To build chemical models, run QSAR predictions, or integrate chemical attributes into machine-learning systems.
Strengths of the PubChem Database (From a Practitioner’s Perspective)
1. Completely Free and Open Access in the PubChem Database
No paywalls, no login required.
This democratized chemical information long before “open science” became a trend.
2. Built on Trusted Sources
NIH, EPA, FDA, CDC, academic research labs, international agencies, and industry contributors.
3. Interlinked Biological and Clinical Data in the PubChem Database
You can move seamlessly from a chemical structure to bioassay results to FDA drug labels.
4. Exceptional Scale
It is, by far, the largest open chemical database in the world.
5. Transparent Data Sources in the PubChem Database
Every data point lists its source—critical for regulatory or scientific work.
Limitations of the PubChem Database You Should Know
PubChem is excellent, but not perfect.
- Not all chemicals have complete experimental property datasets.
- Some data—especially spectral information—may come from older records.
- PubChem aggregates from many sources, so terminology sometimes varies.
- Regulatory classifications differ across countries and may not always align.
For critical safety or legal decisions, PubChem should be paired with primary regulatory sources (OSHA, EPA, NIOSH, FDA).
Practical Tips for Using the PubChem Database Effectively
1. Always Check the Source List
Located at the bottom of each record—this tells you where the information originally came from.
2. Use the “Download” Feature
You can export structures in .sdf, .mol, .csv, .xml, and other formats.
3. Compare Multiple Substance Records
If data differs between providers, this can indicate:
- multiple grades of a chemical
- manufacturing differences
- measurement variability
4. Use Filters to Narrow Down BioAssay Data
Many compounds have hundreds or thousands of test results; filters save hours.
5. Explore “Related Compounds”
This is incredibly useful for drug design and material-science research.
Conclusion: Why the PubChem Database Still Matters
The PubChem database has been a great help to students, scientists, regulators, and even ordinary people. It has converted an enormously complicated issue—chemical information—into something that can be accessed, traced, and used in very practical ways. The very thoroughness of its records, the trustworthiness of its sources, and the openness of its access model make it indispensable to today’s science.
No matter if you are doing a compound verification prior to its synthesis, a toxicity check before a field experiment, or just trying to get a clearer picture of the substances present in a consumer product—PubChem is a very powerful tool that not only should be but also is in fact the very core of every researcher’s work.
If you’re exploring broader public-health data, you can also check our Health Databases section and our guide to the Chemical Contaminants Transparency Tool for deeper context.
Sources Used for the PubChem Database Guide
- PubChem / NCBI — About PubChem
https://pubchem.ncbi.nlm.nih.gov/docs/about - U.S. National Library of Medicine (NIH) – PubChem data ecosystem
https://www.ncbi.nlm.nih.gov/ - FDA DailyMed drug labeling database
https://dailymed.nlm.nih.gov/dailymed/ - EPA CompTox Chemicals Dashboard (regulatory cross-reference)
https://comptox.epa.gov/dashboard
This article was created with AI assistance and reviewed by a human editor.